Revolutionizing prenatal genetic screening
Non-invasive Prenatal Testing (NIPT) is an increasingly common genetic screening performed as early as 9 weeks into pregnancy without risk of miscarriage or harm to the fetus. NIPT requires a liquid biopsy blood sample from the expecting mother. The blood sample contains cell-free DNA (cfDNA), of which 3-13% is fetal cfDNA passed into the mother’s bloodstream through the placenta.1 After extraction and processing, the fetal cfDNA can then be analyzed using next generation sequencing technology to determine risk of the most common chromosome aneuploidies, including Down Syndrome, Edwards Syndrome, and Patau Syndrome as well as chromosome microdeletions and the predicted sex.2
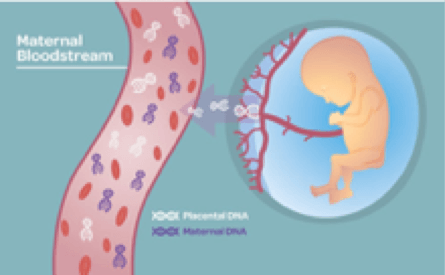
NIPT has been demonstrated to detect genetic abnormality risk with greater accuracy than ultrasound results or family history information, delivering valuable insights on the health of the pregnancy to inform future treatment plans. Particularly for pregnancies with high risk or complications, NIPT provides parents and clinicians with a safe and simple method for early detection and management of expected fetal disorders before birth.3
While highly accurate, NIPT technology does not test for all possible genetic mutations. Further improvements in testing and database development is needed to fully determine the diagnostic capabilities of NIPT.4
The nRichDX Revolution System has the potential to advance NIPT diagnostic precision through increased cfDNA yield, providing testing labs with higher confidence results, and a more complete risk assessment analysis for fetal genetic abnormalities and diseases.
- American College of Obstetricians and Gynecologists. "Committee opinion no. 640: cell-free DNA screening for fetal aneuploidy." Obstet Gynecol 126.3 (2015): e31-e37.
- https://myriadwomenshealth.com/provider/prequel-prenatal-screen/
- https://www.natera.com/vistara
- Lockwood, Christina M. “Unexpected Challenges in Noninvasive Prenatal Testing.” (2015): 1551-1552.

