Revolutionizing early-stage cancer detection
Cell-free DNA (cfDNA) are short, double-stranded DNA sequences that are found circulating in the blood. cfDNA abundance is highly variable, although it can be found in various bodily fluids including blood, urine, lavage, and sputum. While the presence of cfDNA in the body is not inherently indicative of disease, cancer patients typically have higher levels of cfDNA in comparison to healthy individuals.1
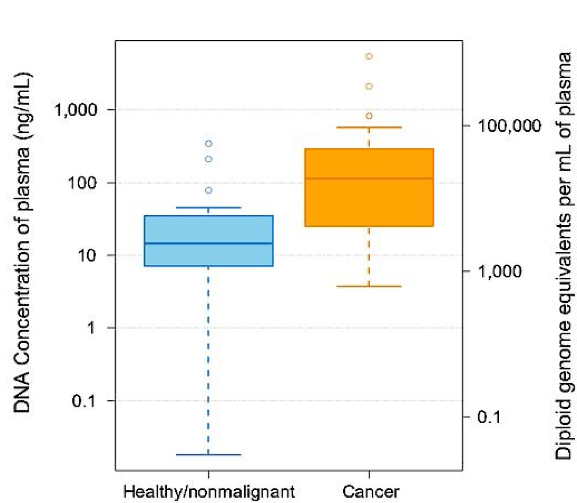
The cfDNA of cancer patients contains circulating tumor DNA (ctDNA) from degraded tumor cells, providing clinicians and researchers with a valuable diagnostic opportunity to profile cancer tumors using liquid biopsy testing. The ability to assess tumor heterogeneity, as well as ease of sampling, minimal invasiveness, and low cost have made liquid biopsy an attractive alternative to traditional (surgical) biopsy-based testing.
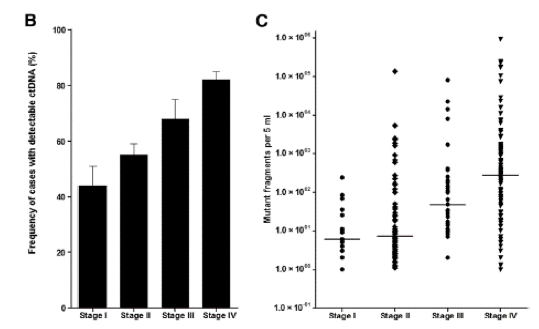
It is known that early-stage tumors produce less ctDNA than more advanced metastic disease.2 As tumor-derived DNA usually constitutes only a small percentage of a patient’s total cfDNA, it is essential that cfDNA analysis testing platforms are sensitive enough to detect rare cancer mutations amidst a wildtype background.
The nRichDX Revolution System greatly improves the chances of recovering these rare mutations amidst cfDNA. The patented process captures more actionable cfDNA by combining increased recovery efficiency with the use of larger sample volume. This combination enables analyte capture that is orders of magnitude more efficient than other sample preparation methods.
Revolution cfDNA Extraction Kit Now Available
Take me to the Revolution cfDNA Extraction Kit product webpage (please scroll to the lower half of the page).
- Volik, Stanislav, et al. "Cell-free DNA (cfDNA): clinical significance and utility in cancer shaped by emerging technologies."Molecular Cancer Research14.10 (2016): 898-908.
- Bettegowda, Chetan, et al. "Detection of circulating tumor DNA in early-and late-stage human malignancies."Science translational medicine6.224 (2014): 224ra24-224ra24.

